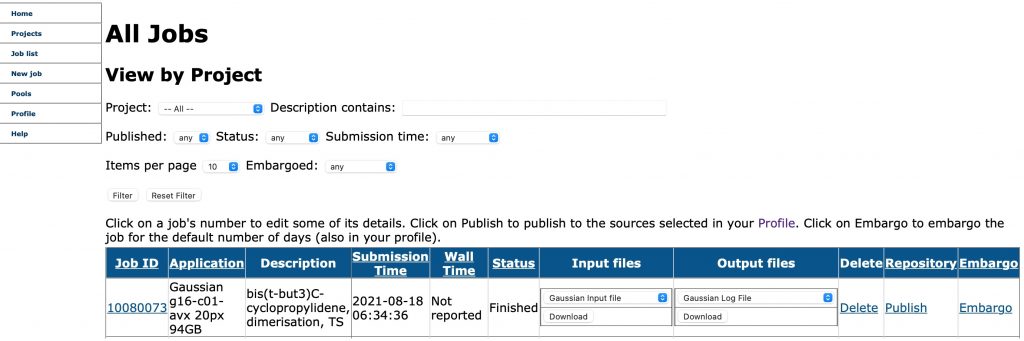
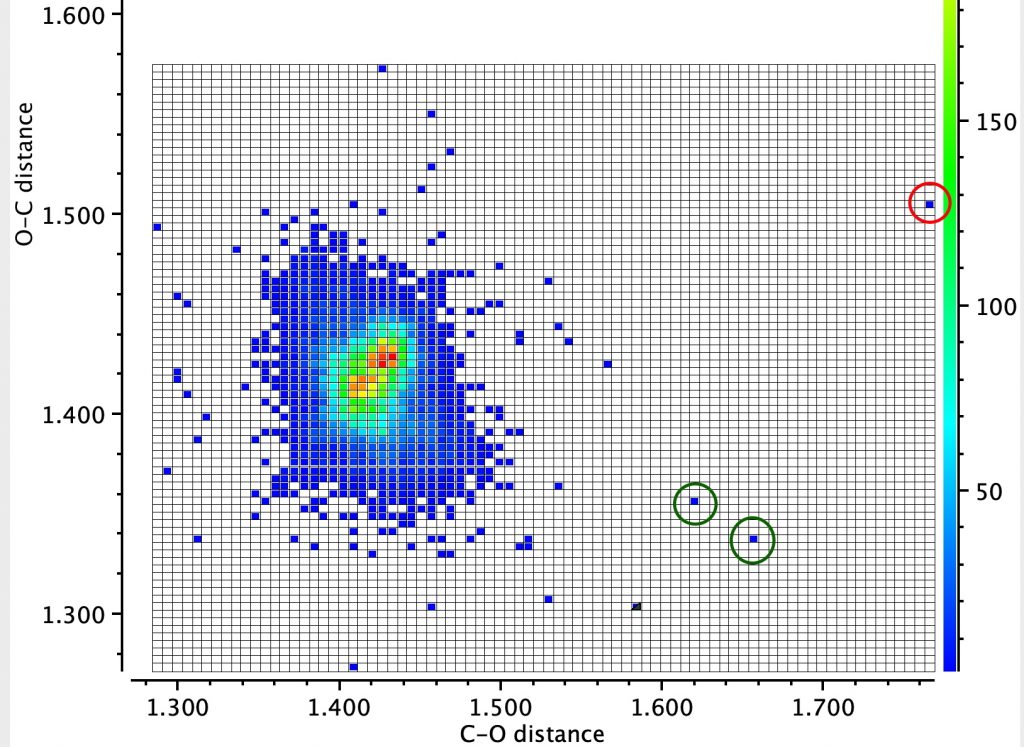
The number of repositories which accept research data across a wide spectrum of disciplines is on the up. Here I report the results of conducting an experiment in which chemical modelling data was deposited in six such repositories and comparing the richness of the metadata describing the essential properties of the six depositions. The repositories are as follows: Figshare as a repository dates from 2012.