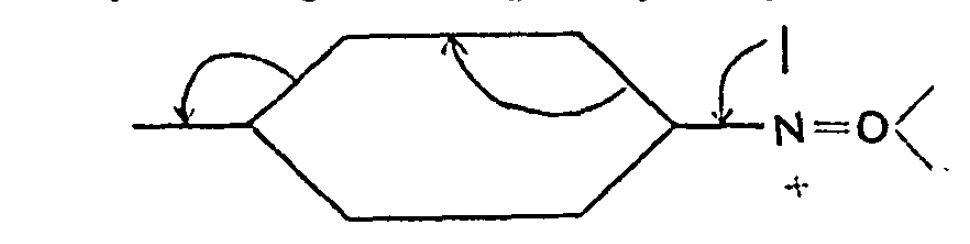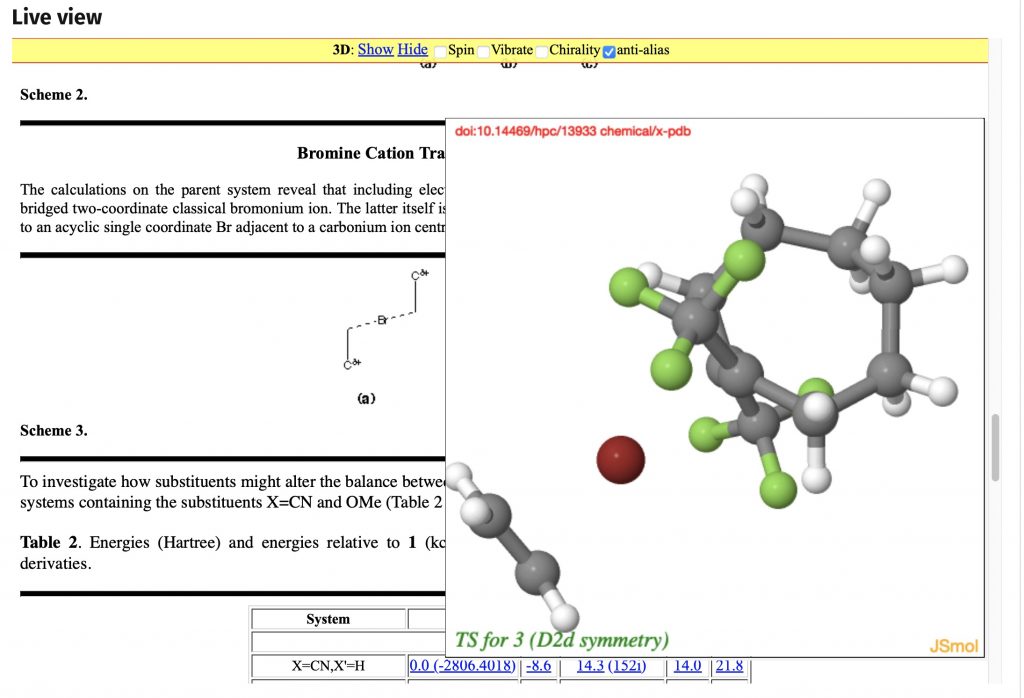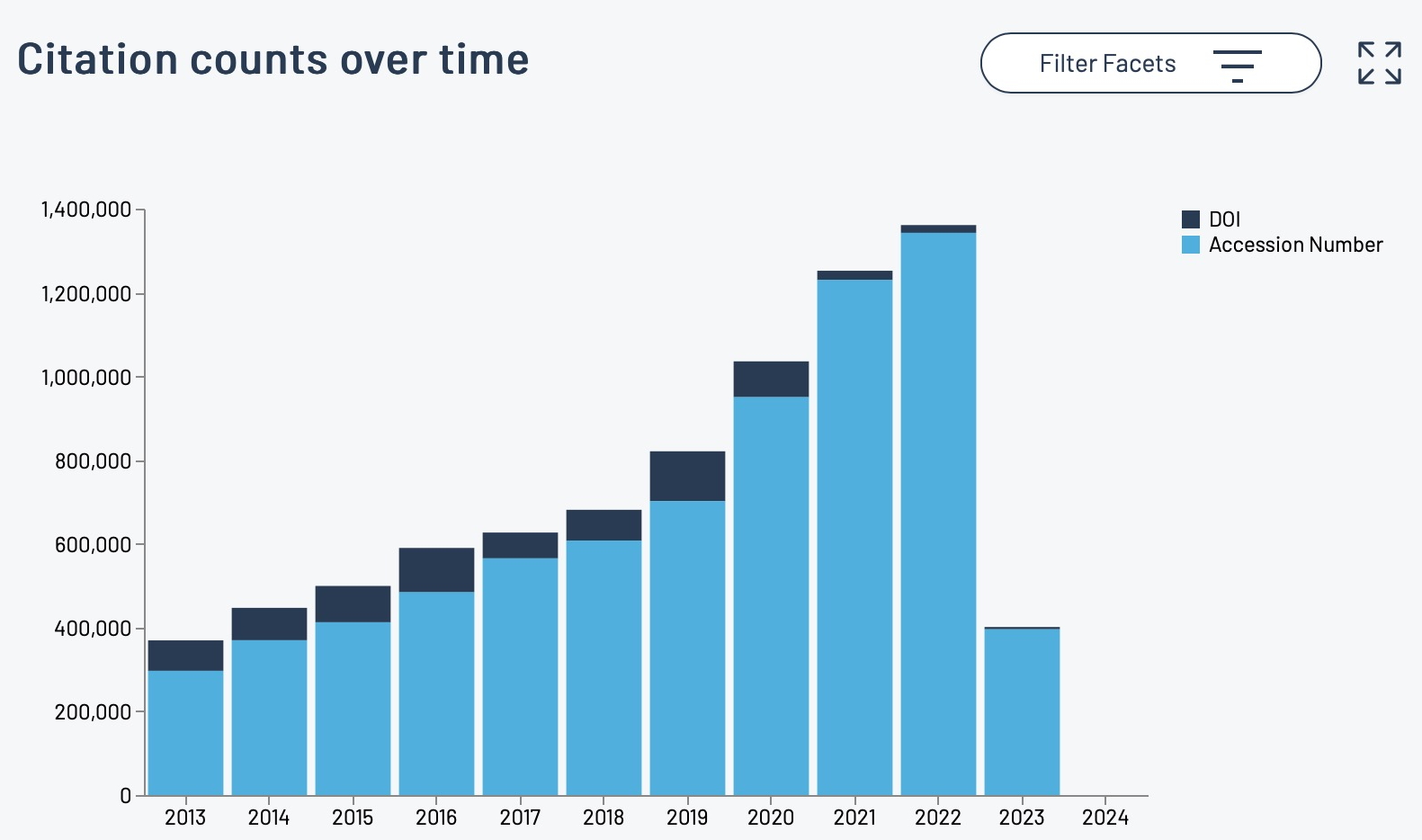
I should start by saying that the server on which this blog is posted was set up in June 1993. Although the physical object has been replaced a few times, and had been “virtualised” about 15 years ago, a small number of the underlying software base components may well date way back, perhaps even to 1993.