
Steve Bachrach has just blogged on a recent article (DOI: 10.1002/anie.200903969) claiming the isolation of a compound with a C≡S triple bond;

Steve Bachrach has just blogged on a recent article (DOI: 10.1002/anie.200903969) claiming the isolation of a compound with a C≡S triple bond;

The Grignard reaction is encountered early on in most chemistry courses, and most labs include the preparation of this reagent, typically by the following reaction:

The so-called Fine tuned model of the universe asserts that any small change in several of the dimensionless fundamental physical constants would make the universe radically different (and hence one in which life as we know it could not exist). I suggest here that there may be molecules which epitomize the same principle in chemistry.
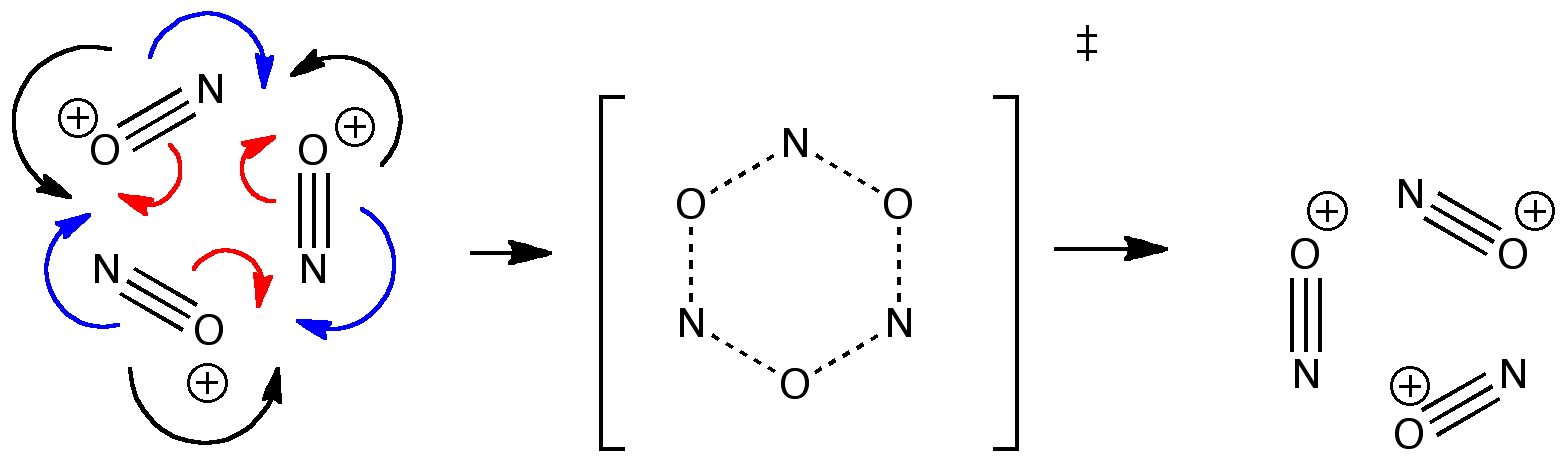
Curly arrow pushing is one of the essential tools of a mechanistic chemist. Many a published article will speculate about the arrow pushing in a mechanism, although it is becoming increasingly common for these speculations to be backed up by quantitative quantum mechanical and dynamical calculations.

In an earlier post I wrote about the iconic SN1 solvolysis reaction, and presented a model for the transition state involving 13 water molecules. Here, I follow this up with an improved molecule containing 16 water molecules, and how the barrier for this model compares with experiment.

We have seen in the series of posts on the topic of hypervalency how the first row main group elements such as Be, B, C and N can sustain apparent hypercoordination and arguably hypervalency.

Quite a few threads have developed in this series of posts, and following each leads in rather different directions. In this previous post the comment was made that coordinating a carbon dication to the face of a cyclopentadienyl anion resulted in a monocation which had a remarkably high proton affinity.

The previous posts have seen how a molecule containing a hypervalent carbon atom can be designed by making a series of logical chemical connections.

The previous post talked about making links or connections.

Science is about making connections. And these can often be made between the most unlikely concepts. Thus in the posts I have made about pentavalent carbon, one can identify a series of conceptual connections. The first, by Matthias Bickelhaupt and co, resulted in the suggestion of a possible frozen SN2 transition state.

In the previous two posts, I noted the recent suggestion of how a stable frozen SN2 transition state might be made. This is characterised by a central carbon with five coordinated ligands.