tpap[cite]10.1055%2Fs-1994-25538[/cite], as it is affectionately known, is a ruthenium-based oxidant of primary alcohols to aldehydes discovered by Griffith and Ley.
I have written earlier about dihydrocostunolide, and how in 1963 Corey missed spotting the electronic origins of a key step in its synthesis.[cite]10.1021/ja00952a037[/cite]. A nice juxtaposition to this failed opportunity relates to Woodward’s project at around the same time to synthesize vitamin B12. The step in the synthesis that caused him to ponder is shown below.
The Sharpless epoxidation of an allylic alcohol had a big impact on synthetic chemistry when it was introduced in the 1980s, and led the way for the discovery (design?) of many new asymmetric catalytic systems. Each achieves its chiral magic by control of the geometry at the transition state for the reaction, and the stabilizations (or destabilizations) that occur at that geometry.
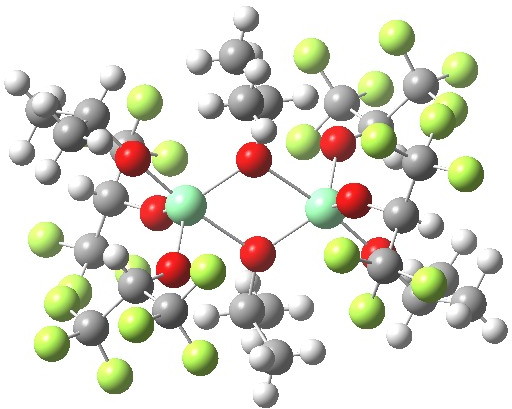
Part one on this topic showed how a quantum mechanical model employing just one titanium centre was not successful in predicting the stereochemical outcome of the Sharpless asymmetric epoxidation.
Sharpless epoxidation converts a prochiral allylic alcohol into the corresponding chiral epoxide with > 90% enantiomeric excess[cite]10.1021/jo00369a032[/cite],[cite]10.1021/jo00360a058[/cite]. Here is the first step in trying to explain how this magic is achieved.
I noted briefly in discussing why Birch reduction of benzene gives 1,4-cyclohexadiene (diagram below) that the geometry of the end-stage pentadienyl anion was distorted in the presence of the sodium cation to favour this product. This distortion actually has some pedagogic value, and so I elaborate this here.
Birch reduction of benzene itself results in 1,4-cyclohexadiene rather than the more stable (conjugated) 1,3-cyclohexadiene. Why is this?
I promised that the follow-up to on the topic of Birch reduction would focus on the proton transfer reaction between the radical anion of anisole and a proton source, as part of analysing whether the mechanistic pathway proceeds O or M . To add some context, Hammond’s postulate [cite]10.1021/ja01607a027[/cite] states that “ the structure of a transition state resembles that of the species nearest
The Birch reduction is a classic method for partially reducing e.g. aryl ethers using electrons (from sodium dissolved in ammonia) as the reductant rather than e.g. dihydrogen. As happens occasionally in chemistry, a long debate broke out over the two alternative mechanisms labelled O (for ortho protonation of the initial radical anion intermediate) or M (for meta protonation).
A dichotomy is a division into two mutually exclusive, opposed, or contradictory groups. Consider the reaction below*.* The bicyclic pentadiene on the left could in principle open on heating to give the monocyclic [12]-annulene (blue or red) via what is called an electrocyclic reaction as either a six (red) or eight (blue) electron process. These two possibilities represent our dichotomy;
