
Here is an inside peek at another one of Derek Lowe’s 250 milestones in chemistry, the polymorphism of Ritonavir .[cite]10.1023/A:1011052932607[/cite] The story in a nutshell concerns one of a pharma company’s worst nightmares;

Here is an inside peek at another one of Derek Lowe’s 250 milestones in chemistry, the polymorphism of Ritonavir .[cite]10.1023/A:1011052932607[/cite] The story in a nutshell concerns one of a pharma company’s worst nightmares;
The previous posts produced discussion about the dipole moments of highly polar molecules.
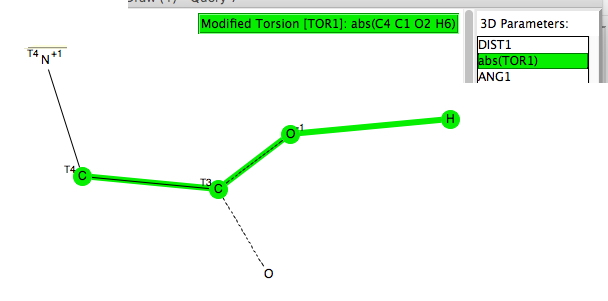
A project fork is defined (in computing) as creating a distinct and separate strand from an existing (coding) project. Here I apply the principle to the polar azulene 4 explored in an earlier post, taking m-benzyne as a lower homologue of azulene as my starting point. m-Benzyne is a less stable 1,3 isomer of o-benzyne (1,2-dehydrobenzene), and is often represented as a 1,3-biradical of 1,3-dehydrobenzene.
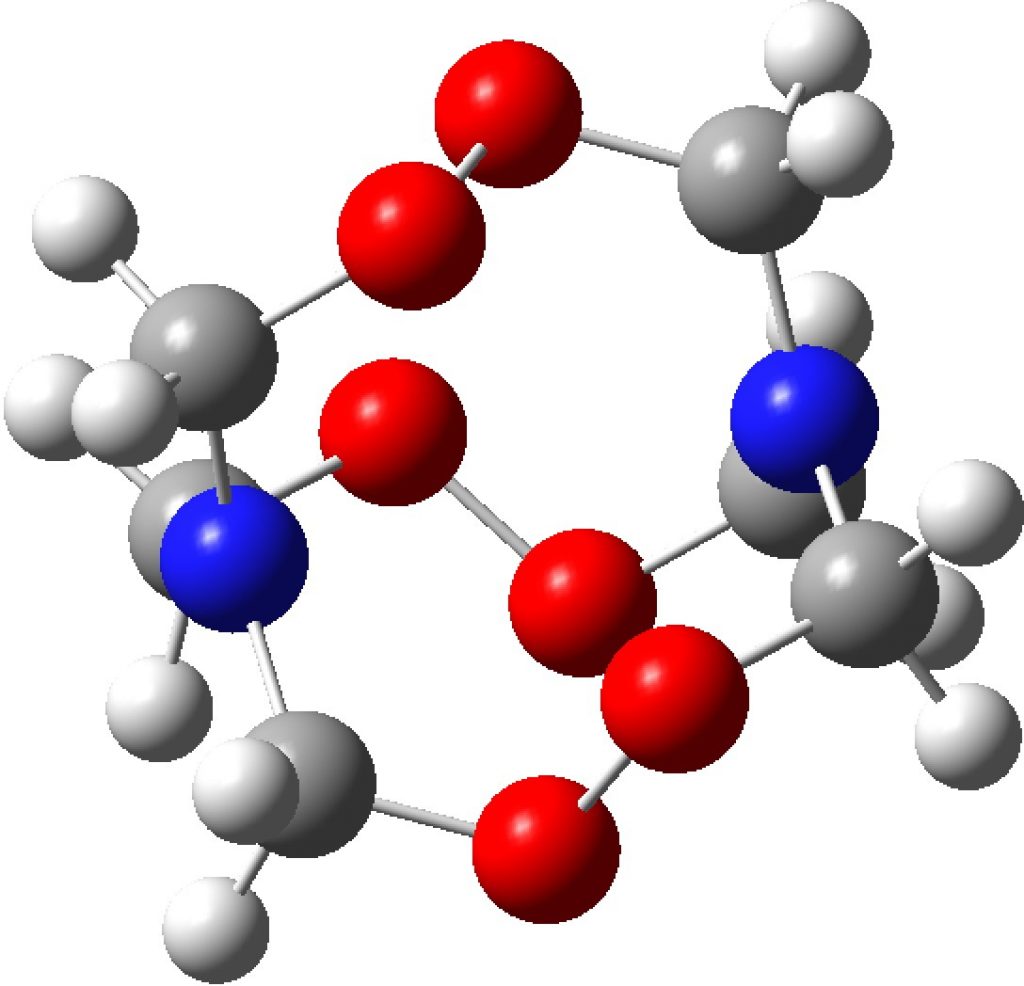
Compounds with O-O bonds often have weird properties. For example, artemisinin, which has some fascinating stereoelectronics. Here is another such, recently in the news and known as HMTD (hexamethylene triperoxide diamine). The crystal structure was reported some time ago[cite]10.1021/jp0123841[/cite] and the article included an inspection of the computed wavefunction.
Bromoallene is a pretty simple molecule, with two non-equivalent double bonds. How might it react with an electrophile, say dimethyldioxirane (DMDO) to form an epoxide?[cite]10.1039/C6CC06395K[/cite] Here I explore the difference between two different and very simple approaches to predicting its reactivity. Both approaches rely on the properties of the reactant and use two types of molecule orbitals derived from its electronic wavefunction.
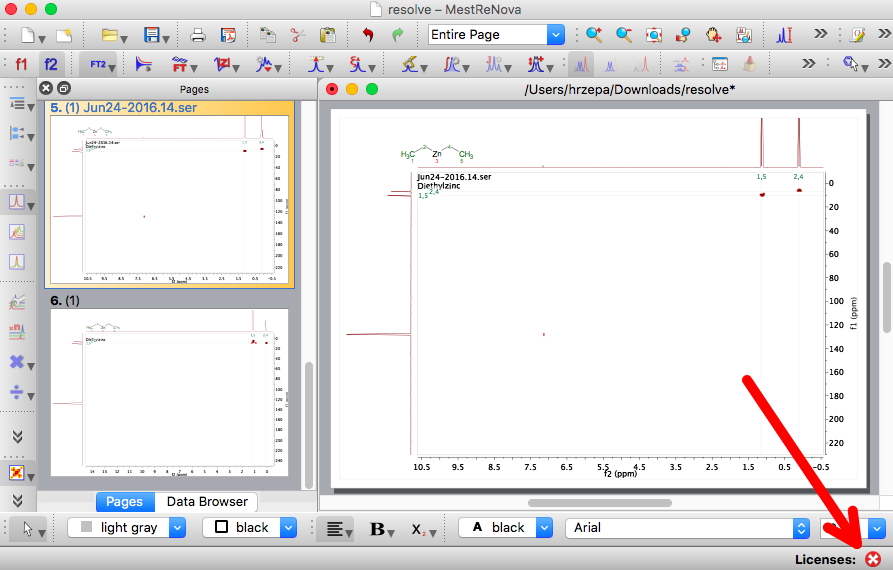
In March, I posted from the ACS meeting in San Diego on the topic of Research data: Managing spectroscopy-NMR, and noted a talk by MestreLab Research on how a tool called Mpublish in the forthcoming release of their NMR analysis software Mestrenova could help. With that release now out, the opportunity arose to test the system.
I have blogged before about the mechanism of this classical oxidation reaction. Here I further explore computed models, and whether they match the observed kinetic isotope effects (KIE) obtained using the natural-abundance method described in the previous post. There is much previous study of this rearrangement, and the issue can be reduced to deciding whether TS1 or TS2 is rate-limiting.
My PhD thesis involved determining kinetic isotope effects (KIE) for aromatic electrophilic substitution reactions in an effort to learn more about the nature of the transition states involved.[cite]10.1039/p29750001209[/cite] I learnt relatively little, mostly because a transition state geometry is defined by 3N-6 variables (N = number of atoms) and its force constants by even more and you get only one or two measured KIE per reaction;
Sodium borohydride is the tamer cousin of lithium aluminium hydride (LAH). It is used in aqueous solution to e.g. reduce aldehydes and ketones, but it leaves acids, amides and esters alone. Here I start an exploration of why it is such a different reducing agent. Initially, I am using Li, not Na (X=Li), to enable a more or less equal comparison with LAH, with water molecules to solvate rather than ether (n=2,3,5) and R set to Me.